Pharmacokinetic analysis normally focuses on systemic exposure to a drug; however, much can be learned from urinary pharmacokinetic parameters. Urinary PK parameters tell you about how much drug was absorbed (at a minimum), and how much drug is eliminated through the kidney. Often it provides easy access to metabolites that are also eliminated in the urine. To understand how to collect urine data and calculate urine PK parameters, it is important to understand what you are measuring.
Urine PK parameters are calculated by relating the amount of drug eliminated in the urine relative to the concentration of drug in the plasma. The bladder is a collection vessel, therefore drug that is eliminated from the body through the kidney collects in the bladder until it is voided from the body with the urine. Thus the concentration of drug in the urine changes with time, and is largely irrelevant. However, the amount of drug in the urine is constantly increasing until all drug is eliminated. Because of this we measure the amount of drug in the urine over a time period rather than the concentration in a single sample.
To properly calculate the amount of drug in a urine sample, you will need to collect all of the urine in an interval following dose administration. Collection intervals normally range from 4 hours to 24 hours. Shorter intervals are used immediately after dosing, and longer intervals are used after a majority of drug has been eliminated. A common sample collection scheme for urine is 0-4 hr, 4-8 hr, 8-12 hr, 12-18 hr, 18-24 hr, 24-48 hr, 48-72 hr. For each collection interval, the following information is required:
- The volume of urine collected. This can be directly measured, or calculated based on the mass of urine, and an average density of urine (1.01 g/mL) using the equation
- The concentration of urine in each collection interval sample. Once the volume is determined, only a small aliquot is needed for the drug concentration assay. Normally 1 – 10 mL of urine is retained for analysis.
- The start and stop time of each interval is required. Timing to the minute is preferred, and patients should try to void at the end of each interval.
After the data is collected, the amount of drug in each collection interval can be calculated using the following equation:
The cumulative amount of drug excreted after each collection interval is then plotted against the median of the collection interval. Nominal or actual times can be used as appropriate. An example data set is shown below:
| Collection Interval (h) | Midpoint for Plots (h) | Volume (mL) | Concentration (ng/mL) | Amount excreted (μg) | Cumulative amount excreted (μg) |
|---|---|---|---|---|---|
| 0 – 4 | 2 | 400 | 111 | 44.4 | 44.4 |
| 4 – 8 | 6 | 290 | 91.3 | 26.5 | 70.9 |
| 8 – 12 | 10 | 390 | 40.3 | 15.7 | 86.6 |
| 12 – 18 | 15 | 460 | 27.2 | 12.5 | 99.1 |
| 18 – 24 | 21 | 415 | 13.8 | 5.7 | 104.8 |
| 24 – 48 | 36 | 1720 | 2.69 | 4.6 | 109.4 |
| 48 – 72 | 60 | 2020 | 0.1 | 0.2 | 109.6 |
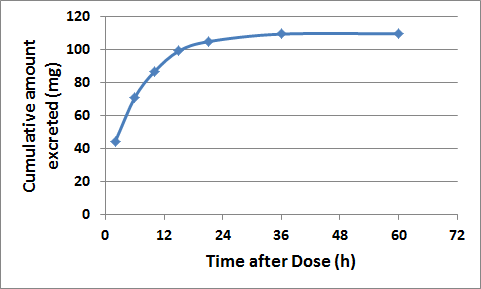
Cumulative amount of drug excreted in the urine
As you can see the drug reaches an asymptote as the elimination of drug slows at around 24-hours after the dose. The asymptote is called the total amount excreted (Ae∞). You can also calculate the renal clearance (CLR) using the following equation:
You can also estimate the elimination half-life of the drug by linear regression of the semi-logarithmic plot of the rate of excretion versus the midpoint of the urine collection time. This is based on the following equations:
There are additional ways to calculate a variety of urinary PK parameters, but these are the basics. I hope that this is helpful!
Today’s pharmacokineticists and PK/PD modelers are under more pressure than ever to quickly and accurately characterize the safety and efficacy profiles of investigational drugs. They need the right tools to perform non-compartmental analysis (NCA), build pharmacometric models, and generate reports that communicate their findings.
Phoenix 7.0’s new features and enhancements are the direct result of user feedback we received to make the world’s most advanced PK/PD software package even better.
Learn more about Certara Phoenix™
Phoenix™ は、PK/PD モデリングに必要な包括的なツールを提供し、前臨床研究者や臨床薬理学者が、複雑な治療法においても自信を持って医薬品開発の意思決定を行えるよう支援します。




