Dose proportionality is a common phrase used pharmacokinetics. Early in the pre-clinical development process, we evaluate dose proportionality in animal species. Then if the drug advances to clinical trials, one of the first assessments in humans is to evaluate dose proportionality. Why is it so important? What do we learn from understanding dose proportionality? What does it mean?
Dose proportionality occurs when increases in the administered dose are accompanied by proportional increases in a measure of exposure like AUC or Cmax. Thus an evaluation of dose proportionality usually includes exposure analysis of three or more doses to produce a graph similar to the one below:
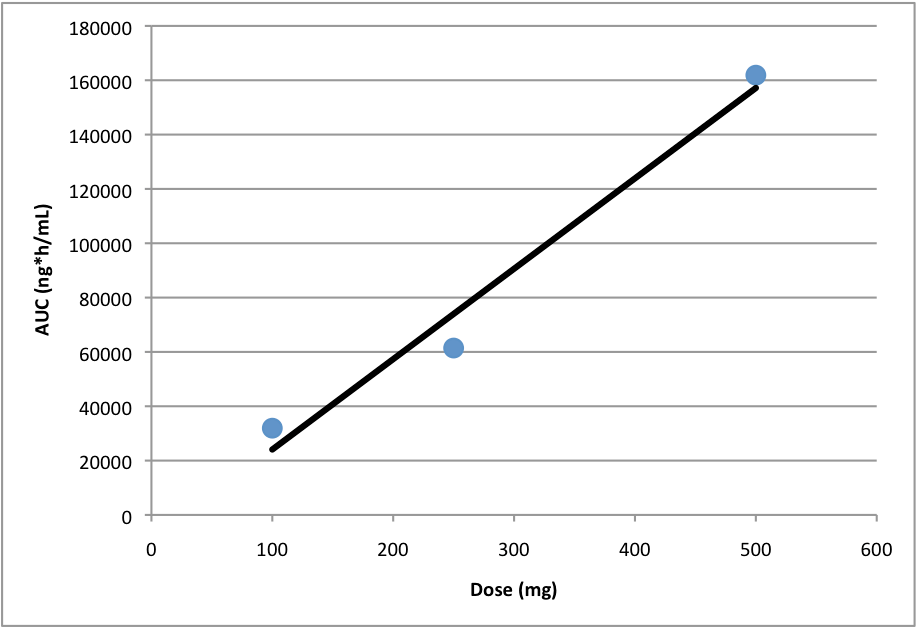
Example of dose proportionality
As shown, a linear regression (or other statistical construct) is used to determine if there is a linear increase in exposure with increases in dose. In general, there are three possible outcomes:
- Exposure is dose proportional (as shown in the figure)
- Exposure is less than dose proportional (points would fall below the line)
- Exposure is greater than dose proportional (points would fall above the line)
The explanation of dose proportionality does little to explain what it means. To understand the utility of dose proportionality, we need to look at the equation for clearance (see previous post):
Equation 1
By rearranging this equation we get the following:
Equation 2
This equation suggests that AUC is linearly related to dose. The proportionality factor is 1/CL. Thus if exposure is dose proportional, it means that clearance is constant over that same dose interval. This is the reason that it is important to test for dose proportionality.
So when testing dose proportionality for your next drug, remember that you are really testing to see if there is constant clearance.
Every patient is different. Thus, they react to drugs in different ways. Precision dosing is a key step toward achieving the goals of precision medicine, a global objective supported by world leaders. The emerging precision dosing field harnesses the explosion of genomic data and various markers of bodily functions using mathematical modeling to ensure that individuals get the best possible treatment.



